The blue and red bow tied in the middle of the rope, pulled by the two creatures represents-the shared pair of electrons-a single bond. The little bunny represents a Hydrogen atom. One example of molecules forming weak bonds with each other as a result of an unbalanced electrostatic potential is hydrogen bonding, where a hydrogen atom will interact with an electronegative hydrogen, fluorine, or oxygen atom from another molecule or chemical group.Īs you can see from the picture above, Oxygen is the big buff creature with the tattoo of "O" on its arm. This potential will make the resulting molecule slightly polar, allowing it to form weak bonds with other polar molecules. As a result of polar covalent bonds, the covalent compound that forms will have an electrostatic potential. A polar covalent bond will result in the molecule having a slightly positive side (the side containing the atom with a lower electronegativity) and a slightly negative side (containing the atom with the higher electronegativity) because the shared electrons will be displaced toward the atom with the higher electronegativity. As a result, the shared electrons will be closer to the atom with the higher electronegativity, making it unequally shared. 
The atom with the higher electronegativity will have a stronger pull for electrons (Similiar to a Tug-O-War game, whoever is stronger usually wins). This occurs when one atom has a higher electronegativity than the atom it is sharing with. It wants to be like Argon who has a full outer valence shell.Ī Polar Covalent Bond is created when the shared electrons between atoms are not equally shared. Phosphorus needs to gain 3 electrons to fulfill the Octet Rule. Argon has a total of 8 electrons (bolded in red), which satisfies the Octet Rule. Although it is important to remember the "magic number", 8, note that there are many Octet rule exceptions.Įxample: As you can see from the picture below, Phosphorus has only 5 electrons in its outer shell (bolded in red). Each atom wants to become as stable as the noble gases that have their outer valence shell filled because noble gases have a charge of 0. It requires 8 electrons because that is the amount of electrons needed to fill a s- and p- orbital ( electron configuration) also known as a noble gas configuration. For Covalent bonds, atoms tend to share their electrons with each other to satisfy the Octet Rule. The Octet Rule requires all atoms in a molecule to have 8 valence electrons-either by sharing, losing or gaining electrons-to become stable. This makes it useful as a base for solder.\) Lead, for example, has such a low melting point that it's easily liquefied by a flame. Melting and boiling points in the carbon family tend to decrease moving down the group, mainly because atomic forces within the larger molecules are not as strong. Group 14 (carbon family) elements have much higher melting points and boiling points than the group 13 elements.Lead is only found as a dense blue-gray metal. Tin occurs as white tin, gray tin, and rhombic tin. Carbon, for example, occurs in diamond, graphite, fullerene, and amorphous carbon allotropes. Except for lead, all of the carbon family elements exist as different forms or allotropes.The elements tend to form covalent compounds, though tin and lead also form ionic compounds.Overall, the carbon family elements are stable and tend to be fairly unreactive.The carbon family elements have widely variable physical and chemical properties. 
Carbon is the only element in the group that can be found pure in nature. These elements are found in a wide variety of compounds.In other words, the elements gain metallicity moving down the group. 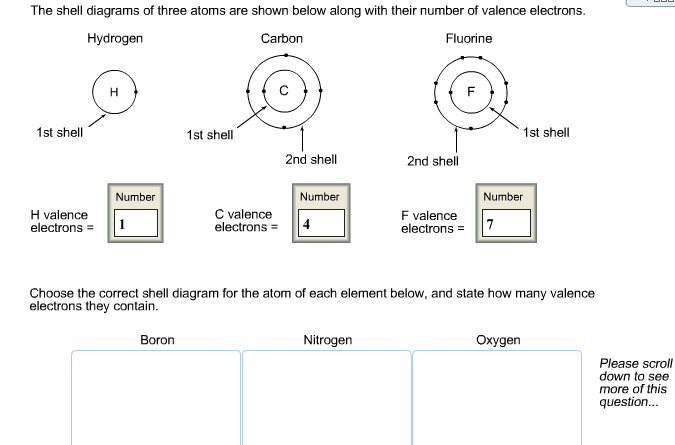
The carbon family consists of one nonmetal (carbon), two metalloids (silicon and germanium), and two metals (tin and lead).Element density increases moving down the group.As you move down the periodic table in the carbon family, the atomic radius and ionic radius increase while electronegativity and ionization energy decrease. Atom size increases moving down the group because an additional electron shell is added.Only carbon has the s 2 outer configuration, which accounts for some of the differences between carbon and other elements in the family. Two of these electrons are in the s subshell, while 2 are in the p subshell. Carbon family elements contain atoms that have 4 electrons in their outer energy level.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
